Plasma Medicine
High-performance mass spectrometry for medical plasma characterization
Hiden Analytical MBMS (molecular beam mass spectrometry) systems are optimized to accurately analyze hydrated cluster species and reactive oxygen and nitrogen species from atmospheric cold plasma treatment systems.
Related Products
Plasma medicine is an innovative and interdisciplinary field that focuses on using ionized gas (plasma) for medical applications such as disinfection, wound healing, and cancer treatment.1-3
Applications of plasma medicine can be broadly categorized into three groups:
- Direct therapeutic application of atmospheric-pressure plasma in vivo
- Plasma-assisted preparation of biocompatible surfaces
- Plasma decontamination and sterilization in medical environments
The fundamental enabler of in vivo therapeutic plasma medicine is the production of ‘cold plasma’ at atmospheric pressure. A cold (or nonthermal) plasma consists of ionized gas in which the dissociated electrons have a high temperature, while the ions and neutral atoms remain only slightly above room temperature. Producing cold plasma at atmospheric pressure facilitates its direct application to living tissue, at which point it can selectively kill or inactivate microorganisms without heating or damaging surrounding tissues. A range of other plasma-cell and plasma-tissue interactions can also be produced, including selective inactivation or destruction of mammalian cells and detachment of cells from matrices and cell clusters.4
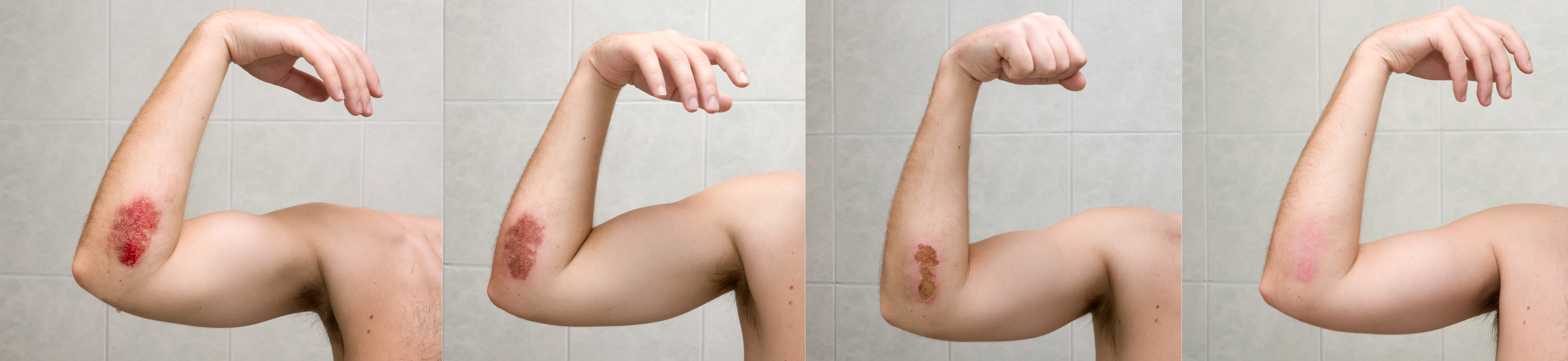
Cold Atmospheric Plasmas for Wound Healing
At present, this research’s primary focus is on wound healing and the treatment of pathogenic skin diseases. Atmospheric cold plasma can reduce bacterial load on wound sites while stimulating tissue regeneration, enabling acceleration of the healing process.
The mechanisms of plasma-cell and plasma-tissue interactions are complex and are the subject of a growing body of recent research. However, it has been established that significant biological plasma effects are caused by plasma-induced changes to the liquid environment of cells and tissues and by reactive oxygen species (ROS) and reactive nitrogen species (RNS) present in the plasma.
A range of very different experimental setups and plasma sources have been used for medical plasma research. Many plasma sources have tunable parameters such as power, gas species, and gas flow rate, allowing the effects of various plasma modifications to be studied.
The HPR-60 molecular beam mass spectrometer has been developed specifically for the analysis and characterization of atmospheric plasmas. The three-stage skimmer cone configuration enables pressure reduction to ultra-high vacuum (UHV) for mass and energy analysis and the creation of a supersonic ‘silent’ region behind the first skimmer cone, preventing further collisions and interactions of the species sampled by the MS system.
The composition and quantity of plasma-generated ROS and RNS depend on specific plasma source and device parameters and working gas composition, power input, and distance from the plasma source. The HPR-60 enables direct and efficient characterization of the effects of these parameters on plasma properties.
The HPR-60 system can be equipped with the EQP or EPIC ranges of Hiden mass spectrometers. These support mass options up to 5000 amu – making the system ideal for analyzing hydrated cluster species that have been found to form in atmospheric plasma processes. For more information on our plasma analysis and mass spectrometry solutions, contact the Hiden Analytical team today.
HPR-60 MBMS – The Ultimate Solution for Reactive Gas and Plasma Analysis
Atmospheric Plasma Applications Catalogue
EQP Series Mass and Energy Analysers
Plasma Etch in Microtechnology
ESPION Advanced Langmuir Probes for Plasma Diagnostics
HPR-60 Molecular Beam Sampling Mass Spectrometer
Mass Spectrometers for Thin Films, Plasma and Surface Engineering
Atmospheric Plasma Analysis by Molecular Beam MS – GEC 2004 (1.38 MB)
Atmospheric Pressure Plasma Analysis by Modulated Molecular Beam MS – ICPIG 2005 (256 KB)
Ion Energy Distributions for a DC Plasma – GEC 2003 (250 KB)
Mass Analysis of CF3I Decomposition in a Surface Barrier Discharge – GEC 2011 (2.8 MB)
Mass Spectroscopy of Metastable Species during Plasma Processing – GEC 2011 (2.1 MB)
Time Resolved Ionisation Studies of HIPIMS – PSE 2006 (848 KB)
- von Woedtke, Th., Metelmann, H.-R. & Weltmann, K.-D. Clinical Plasma Medicine: State and Perspectives of in Vivo Application of Cold Atmospheric Plasma: Clinical Plasma Medicine: State and Perspectives of in Vivo Application of Cold Atmospheric Plasma. Contrib. Plasma Phys. 54, 104–117 (2014).
- Kong, M. G. et al. Plasma medicine: an introductory review. New J. Phys. 11, 115012 (2009).
- Fridman, G. et al. Applied Plasma Medicine. Plasma Processes and Polymers 5, 503–533 (2008).
- von Woedtke, Th., Reuter, S., Masur, K. & Weltmann, K.-D. Plasmas for medicine. Physics Reports 530, 291–320 (2013).

