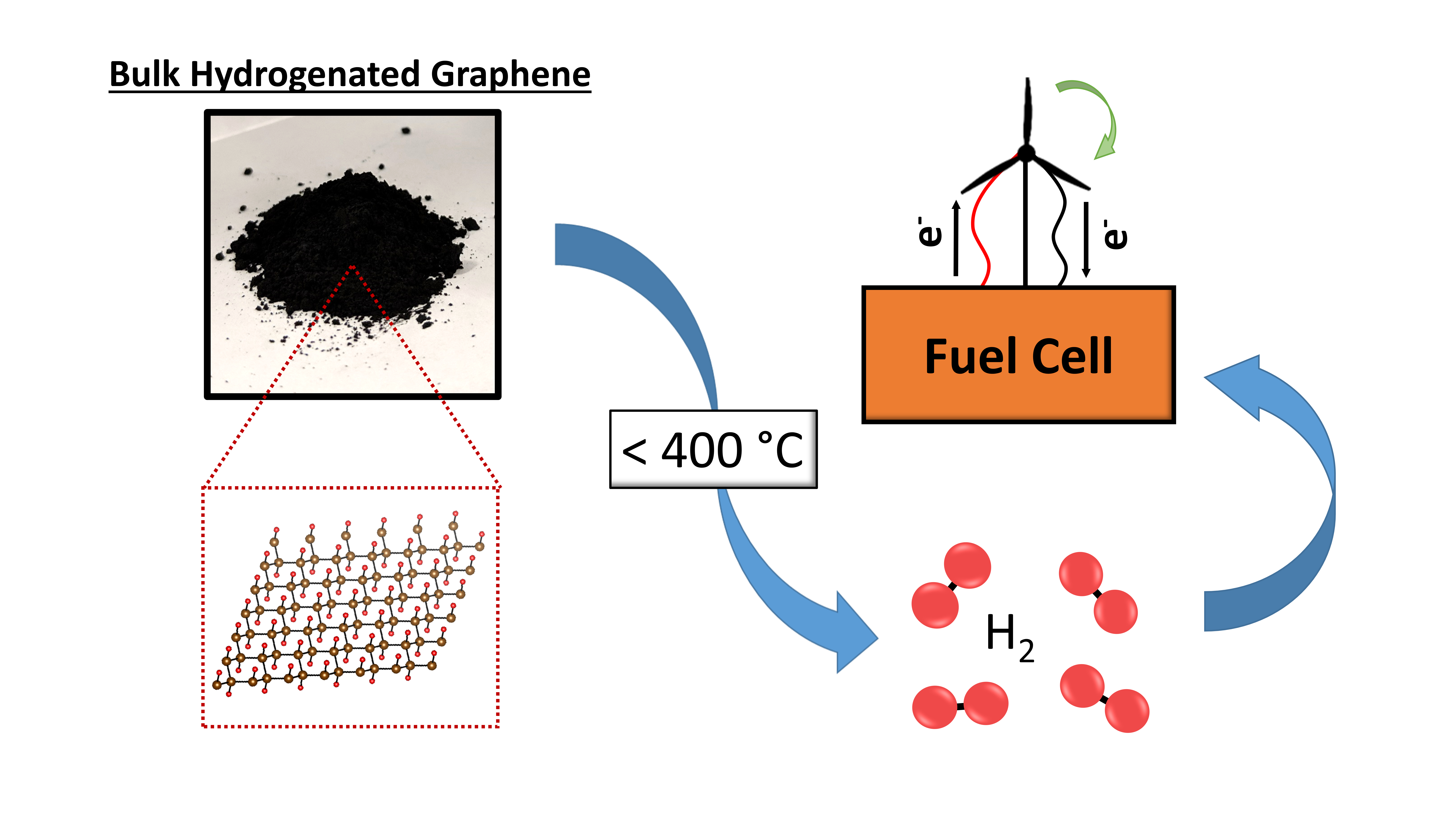
Fully hydrogenated graphene, termed “graphane”, is composed of 7.7 % hydrogen by mass, and possesses unique physical, electrical, and optical properties. Like graphene however, pristine graphane is challenging to isolate in bulk form. However, graphite and similar carbon allotropes can be chemically hydrogenated to yield similar materials which are colloquially termed hydrogenated graphene. These materials are stable under ambient conditions, but will liberate hydrogen gas upon thermal decomposition above 400° C, making them promising materials for hydrogen storage.
Our group is interested in evaluating the viability of hydrogenated graphene as a means of hydrogen storage.1,2 We are currently perusing fundamental studies regarding the material’s scaled up synthesis, characterization and testing. In order to identify the material’s volumetric and gravimetric hydrogen storage capacity, as well as the purity of the evolved hydrogen gas, accurate characterization and quantification of the generated gases are required.
To understand the dynamics of thermal decomposition, hydrogenated graphene was characterized via thermal gravimetric analysis coupled with mass spectrometry. Samples of hydrogenated graphene were heated at a controlled rate with the evolved gases characterized in real time using a Hiden HPR-20 mass spectrometer. Additionally, the quantity and composition of gaseous species generated from the thermal decomposition of hydrogenated graphene samples were identified by decomposing a known amount of hydrogenated graphene in a sealed pressure reactor plumbed to the calibrated Hiden HPR-20 mass spectrometer.
Along with hydrogen gas, both water and ammonia were among the gaseous products observed during decomposition. However, accurate differentiation of water and ammonia species can be challenging by mass spectrometry due to spectral overlap of the two species’ mass fragments.3 Appearance Potential Soft Ionisation (APSI) mode was applied to tune discrete ion energies for individual mass channels to selectively identify the parent ions and minimize spectral overlap. This enabled unambiguous characterization and quantification of all evolved gases, including water and ammonia species, during decomposition of the hydrogenated graphene.
This characterization, along with other complementary techniques,1,2 enabled thorough evaluation of hydrogenated graphene as a means to store and deliver hydrogen gas on demand. Using our synthetic protocol, hydrogenated graphene was found to possess a hydrogen storage capacity of ~3.2 wt.%, which is roughly 42% of the theoretical value of graphane. While improvements to our synthetic protocols are anticipated to further increase the H2 storage capacity of this material (and are currently under way), the results obtained demonstrate multi-gram quantities of hydrogenated graphene can be used to safely generate practical quantities of H2 gas on demand.
 Project summary by: James R. Morse, Naval Research Laboratory, Materials Science & Technology Division, Washington, DC, 20375 USA
Project summary by: James R. Morse, Naval Research Laboratory, Materials Science & Technology Division, Washington, DC, 20375 USA
Paper Reference: “Hydrogenated graphene: Important material properties regarding its application for hydrogen storage” Journal of Power Sources (2021) 494, 229734 doi.org/10.1016/j.jpowsour.2021.229734
References:
- R. Morse, D.A. Zugell, B.R. Matis, H.D. Willauer, R.B. Balow, J.W. Baldwin, Int. J. Hydrogen Energy 45 (2019) 2135.
- Power Sources 494 (2021) 229734.
- “APSI mode simplifies MS spectral analysis” https://www.hidenanalytical.com/wp-content/uploads/2017/06/HAPR0174_APSI-mode-simplifies-MS-spectral-analysis.pdf (accessed Aug. 20, 2021).
Hiden Product: HPR-20
Reference: AP-HPR-20-202147
To find out more about these products visit the HPR-20 product page or if you would like to contact us directly please Send us a Message.

