Investigation of MoOx/Al2O3 under Cyclic Operation for Oxidative and Non-Oxidative Dehydrogenation of Propane
Catalysts 2020, 10, 1370; doi:10.3390/catal10121370
In order to bridge the propene supply and demand gap, the chemical industry has looked to highly selective on-purpose technologies like propane dehydrogenation (DP) and propane oxidative dehydrogenation (ODP). However, both techniques have potential disadvantages. In order to overcome these problems, studies have been conducted into the use of supported molybdenum oxide to enhance selectivity for propylene during ODP and DP reactions. Using the Hiden CATLAB together with an integrated mass spectrometer (QGA), a novel reaction cycle has been investigated and shows an increase in propene formation in the second ODP reaction whilst maintaining a similar propane conversion.
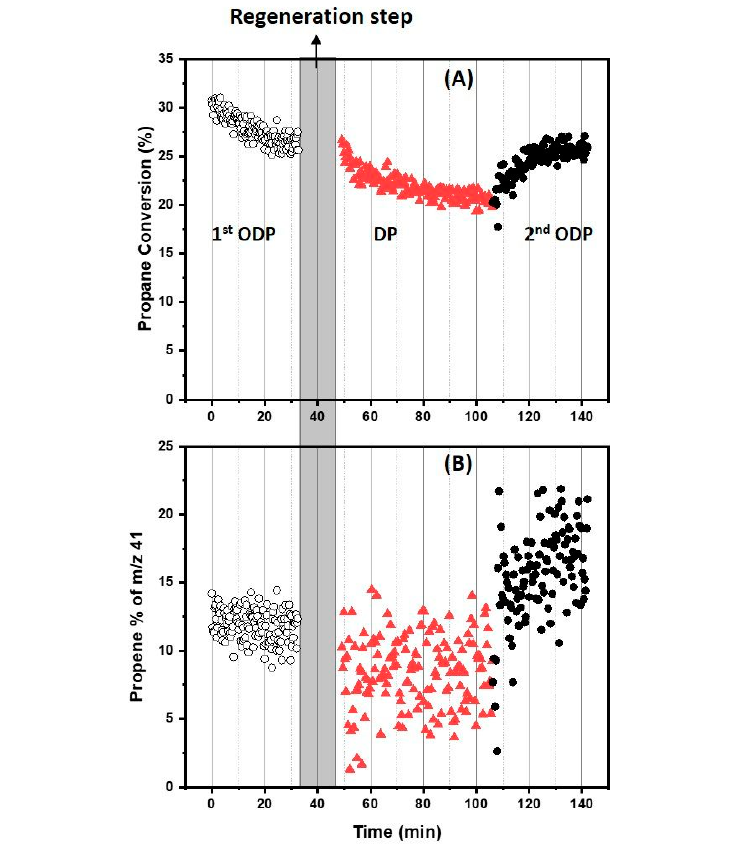
Figure 1. The subsequent oxidative and non-oxidative dehydrogenation of propane activit y over MoOx/Al2O3 at 500 oC shown as (A) Propane conversion and (B) Propene formation. Propene formation is presented as the contribution of propene to the mass spectrometry response of m/z 41 expressed as a%. This is calculated by subtracting the contribution of propane to m/z 41. Refer to the Materials and Methods section for activity test details.
You can Download & read the full paper or View it online.
To find out more about this product visit the CATLAB and QGA product page or if you would like to contact us directly please Send us a Message.

