 Conversion of natural gas or methane to hydrogen and valuable commodities with high efficiency is relentlessly pursued. Compared with the steam reforming of methane, catalytic partial oxidation of methane (CPOM) is a preferred technology to produce hydrogen or synthesis gas with more economic incentives. Supported nickel (Ni) catalysts are widely used for the CPOM due to low cost and high effectiveness as compared to noble metals. However, application of the Ni catalyst to the CPOM is limited due to deactivation of Ni nanoparticles by sintering and coking, i.e., carbon deposition. Our group demonstrates that the microwave-assisted synthesis is a convenient way to make an effective NiO/SDC catalyst that catalyzes partial oxidation of methane toward syngas production. The effect of Ni loadings and Sm/Ce ratios on the CPOM activity are investigated using a Hiden HPR-20. The high activity was attributed to nanoscale particle size, which provokes interaction between Ni and SDC easily. These findings provide information of NiO/SDC catalysts for use in other oxidation reactions and allows better design of other element doped ceria catalysts. Besides, the synthesis approach can be applied to other materials or oxides, where tedious procedures for the synthesis are required.
Conversion of natural gas or methane to hydrogen and valuable commodities with high efficiency is relentlessly pursued. Compared with the steam reforming of methane, catalytic partial oxidation of methane (CPOM) is a preferred technology to produce hydrogen or synthesis gas with more economic incentives. Supported nickel (Ni) catalysts are widely used for the CPOM due to low cost and high effectiveness as compared to noble metals. However, application of the Ni catalyst to the CPOM is limited due to deactivation of Ni nanoparticles by sintering and coking, i.e., carbon deposition. Our group demonstrates that the microwave-assisted synthesis is a convenient way to make an effective NiO/SDC catalyst that catalyzes partial oxidation of methane toward syngas production. The effect of Ni loadings and Sm/Ce ratios on the CPOM activity are investigated using a Hiden HPR-20. The high activity was attributed to nanoscale particle size, which provokes interaction between Ni and SDC easily. These findings provide information of NiO/SDC catalysts for use in other oxidation reactions and allows better design of other element doped ceria catalysts. Besides, the synthesis approach can be applied to other materials or oxides, where tedious procedures for the synthesis are required.
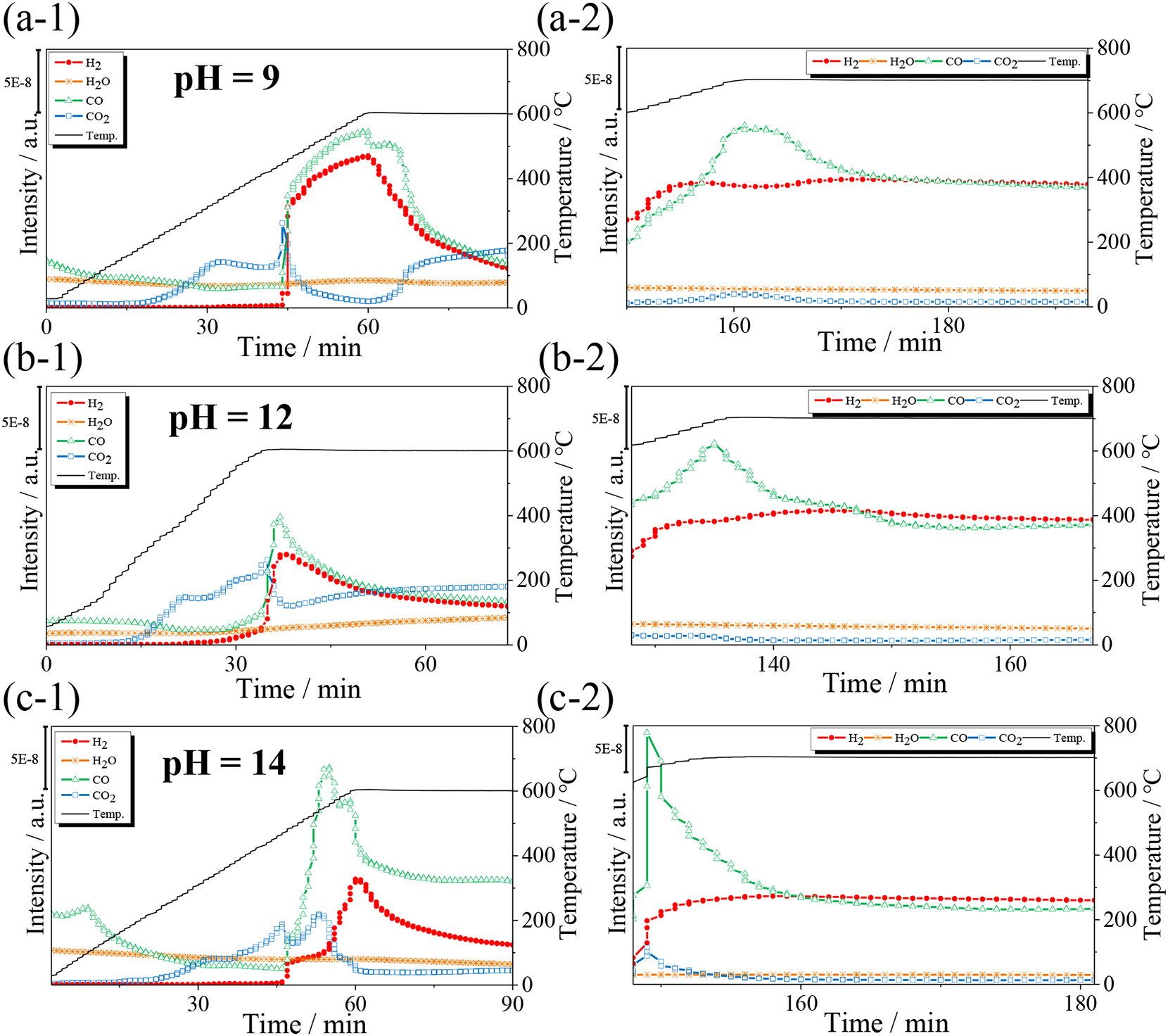
Fig. 4. MS profiles of eluting gas products from methane oxidation on the co-microwave synthesized Ni/SDC catalyst prepared at different pH values, pH = 9 (a1-a2), pH = 12 (b1-b2), and pH = 14 (c1-c2).
 Project summary by: Andrew C. Chein, Department of Chemical Engineering, , Taichung 40724, Taiwan
Project summary by: Andrew C. Chein, Department of Chemical Engineering, , Taichung 40724, Taiwan
Paper Reference: “Effect of preparation method and particle size of Ni/SDC catalyst on methane oxidation” Catalysis Communications (2021) 154, 106312
Hiden Product: HPR-20
Reference: AP-HPR-20-202153
To find out more about these products visit the HPR-20 product page or if you would like to contact us directly please Send us a Message.

